 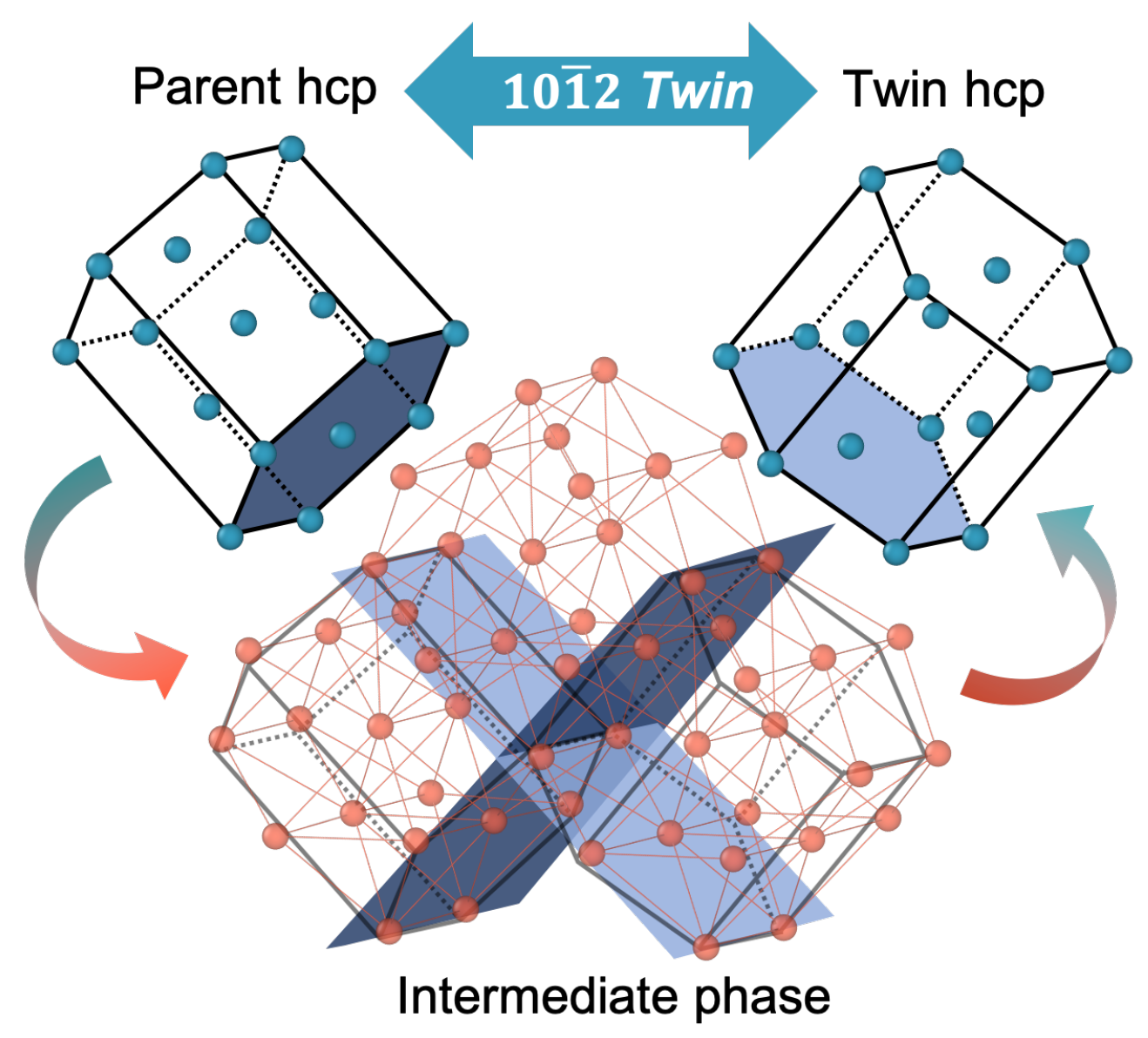
In both these cases, magnesium proves to be a very essential element to the biological component. Without magnesium, plants would not be able to commence the process of photosynthesis, leading to loss of oxygen and an increase in carbon dioxide, and also, the human body would not be able to function efficiently due to lack of enzyme activity. To maintain this ongoing process, the normal human body consumes approximately 250-350 mg of Magnesium everyday and store 20 grams in our body, mainly in the bones. By looking at an atom’s electron configuration, it is possible to determine how many energy levels the atom has, how many electrons are in each energy level and how many electrons are in. The electronic configuration of Magnesium is (2,8,2) -> meaning that there are 2 electrons in the first valance shell, 8 in the second and 2 in the third leaving the third valance shell incomplete. Electron configuration is a representation of how electrons are arranged in an atom or molecule. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. This element is essential to many enzymes and their work. The electron configuration for magnesium is 1s2 2s2 2p6 3s2. Now, let’s begin with drawing the Lewis structure of magnesium fluoride (MgF2): Step 1: Search for the maximum available valence electrons in one magnesium fluoride molecule: It is sixteen as two are with magnesium atom and fourteen are with two fluorine atoms. Not only this, but Magnesium is also an important component in the human body. Here, the valence electrons in fluorine are 9. As chlorophyll has properties of magnesium within it, photosynthesis can not take place without its existence, this would not only cause disturbance to the life of plants but also disrupt the human race and its survival. 12 electrons (white) occupy available electron shells (rings). 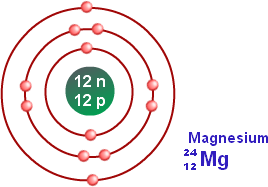
The nucleus consists of 12 protons (red) and 12 neutrons (orange). This lustrous silvery-white metal is not only significant due its metallic appearance, it is also an essential element to both plant and animal life. In 2011, the Commission has changed the standard atomic weight of magnesium to Ar(Mg) 24.304, 24.307 based on an evaluation of the effect of variation. Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of magnesium-24 (atomic number: 12), the most common isotope of this element.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
